Gut Instincts
"In recent years, scientific interest has grown in the connection between the gut and the brain as it used to be a relationship that was overlooked but now it is recognised as critical in both physical and mental health."
Amanda Dias
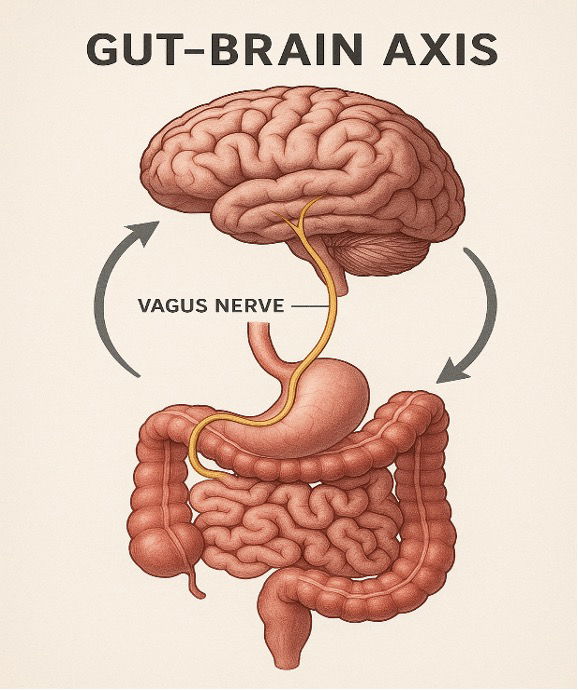
Exploring the Gut-Brain Axis.
How Digestive Health Influences Mental Wellbeing and Therapeutic Interventions.
How Digestive Health Influences Mental Wellbeing and Therapeutic Interventions
In recent years, scientific interest has grown in the connection between the gut and the brain as it used to be a relationship that was overlooked but now it is recognised as critical in both physical and mental health.
The gut is not just a digestive organ, it is a complex and intelligent system that communicates directly with the brain via the vagus nerve. It earned the title of the body’s second brain due to the presence of the Entric Nervous System (ENS), which is a vast network of 500 million neurons embedded in the gut wall.
The ENS can operate independently of the central nervous system and is involved in producing key neurotransmitters like serotonin, which regulates both gut function and mood.
Working alongside the ENS is the gut microbiome where trillions of bacteria live in the intestines, influencing inflammation, stress responses, and even behaviour.
Together, these systems form the gut-brain axis, a bidirectional communication pathway that links digestive and mental health. This project explores how disruptions in this axis, such as in coeliac disease or IBS, can contribute to psychological symptoms like anxiety and depression, and how gut-focused therapies may offer new approaches to treating the mind through the body and how psychological therapies can offer improvements in gut symptoms.(1)
This second brain maintains constant communication with the actual brain through a two-way system known as the gut-brain axis, primarily mediated by the vagus nerve and chemical messengers like neurotransmitters. This ongoing dialogue influences many aspects of our health and well-being. However, when this connection becomes disrupted, the effects can range from mild to severe. For some, it might cause occasional discomfort or bloating - perhaps after indulging in too much junk food - signaling the gut and brain trying to restore balance. For others, the disruption results in intense pain, nausea, and digestive distress that can severely impact daily life. Despite its profound effects, many people remain unaware of this crucial link, leading to misunderstandings and underdiagnosed conditions that affect both physical and mental health.
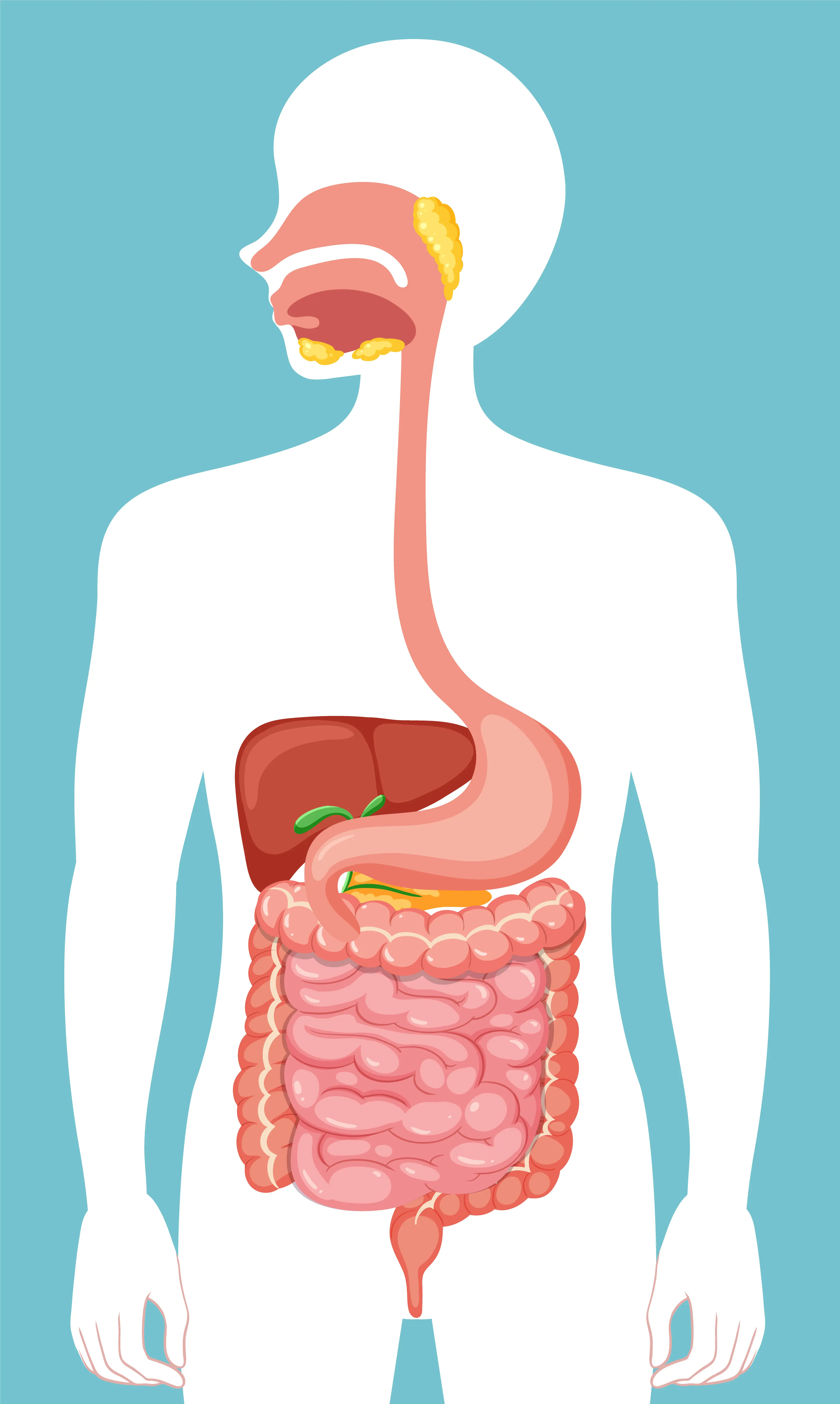
Overview of the Digestive System
The digestive system is far more than just a food-processing machine; it’s a complex, finely tuned network essential to our overall health. As Giulia Enders highlights in her book ‘Gut’, digestion begins the moment food enters our mouths, where enzymes in saliva start breaking it down. From there, food travels down the oesophagus into the stomach, a powerful muscular organ that churns and bathes food in acid, turning it into a semi-liquid mixture called chyme.
The journey continues into the small intestine, a remarkable six-meter-long tube lined with millions of microscopic villi that absorb nutrients into our bloodstream. The pancreas and liver join in, releasing enzymes and bile that help digest fats, proteins, and carbohydrates. Finally, the large intestine (colon) reclaims water and houses the dense, diverse community of microbes known as the gut microbiome.
She also further explains that this system is not merely mechanical: it is deeply connected to our emotions, immune responses, and even decision-making. The gut’s own nervous system allows it to act semi-independently, coordinating digestion while also sending and receiving messages from the brain. This intricate collaboration ensures that digestion is seamlessly integrated with how we feel, behave, and function in daily life.

What is the Gut Microbiome and
What Does It Do?
The gut microbiome is the trillions of bacteria living in our gut that have various functions and contribute to various systems within the human body. Research by Jeffrey I. Gordon has shown that the gut microbiome plays a crucial role in digestion, metabolism, and even mental health. Gordon’s studies revealed that gut bacteria break down complex carbohydrates and fibres that human enzymes cannot digest, producing short-chain fatty acids (SCFAs) and other metabolites. These metabolites act as signalling molecules that help regulate immune responses and support the intestinal barrier by strengthening tight junctions and modulating epithelial cell function, reducing systemic inflammation.(2)
In addition, gut microbes contribute to the production of neurotransmitters such as serotonin and gamma-aminobutyric acid (GABA), which can influence mood and behaviour.(3)
Disruptions in this microbial ecosystem, known as dysbiosis, have been linked to a range of conditions, from obesity and metabolic disorders to anxiety and depression. For instance, about 38 % of people diagnosed with a gastrointestinal disorder like irritable bowel syndrome (IBS) also experience clinically significant anxiety, and around 27 % experience depression, according to a large study of over 1.2 million US hospital admissions between 2016 and 2019.(4)
To understand the causal role of the microbiome, Gordon’s team famously transplanted gut microbes from obese and lean humans into germ-free mice. The mice colonised with microbes from obese donors developed increased fat mass and metabolic changes compared to those that received microbes from lean donors, demonstrating that the microbiome itself can drive differences in metabolism and disease risk.(5)
The Gut-Brain Axis: ENS, Vagus Nerve & Bidirectional Signaling
As previously mentioned, the gut-brain axis is a sophisticated, bidirectional communication network that connects the enteric nervous system (ENS) with the central nervous system, primarily via the vagus nerve. Beyond this neural connection, the axis involves a complex interplay of immune signalling, endocrine pathways, and microbial metabolites, all contributing to the dynamic dialogue between gut and brain.
The ENS, with its extensive neuronal network, acts as an autonomous controller of gastrointestinal function but also constantly integrates signals from the brain to fine-tune digestive processes. The vagus nerve transmits afferent (sensory) signals about gut luminal contents, microbial activity, and inflammation to brain regions responsible for self-awareness, emotional processing, and memory, such as the insula, limbic system and the hippocampus. In return, efferent (motor) signals from the brain modulate gut motility, secretion, and immune responses, maintaining homeostasis.
- STEP 01
Enteric Nervous System
The enteric nervous system is a network of neurons in the gut that controls digestion independently.
- STEP 02
Vagus Nerve
The vagus nerve is a major nerve that carries signals between the brain and vital organs.
- STEP 03
Bidirectional Signaling
Bidirectional signalling is two-way communication between two systems, each influencing the other.
When Things Go Wrong: Coeliac Disease, DGBIs & Gut Dysfunction
The gut-brain axis’s delicate balance can be disrupted by a range of gastrointestinal disorders, with coeliac disease, Disorders of Gut-Brain Interaction (DGBIs), and various food intolerances standing out as key examples of how gut dysfunction can severely impact both digestive and mental health.
Identifying Disorders of Gut-Brain Interaction (DGBIs) remains a major challenge in gastroenterology, as these conditions, which include irritable bowel syndrome and functional dyspepsia, lack clear structural or biochemical markers. Traditionally, diagnosis relies on detailed patient history, symptom-based criteria like the Rome IV, and ruling out organic disease through tests. However, researchers are now exploring more precise tools to detect subtle physiological and biological differences that contribute to symptoms. For example, the search for biomarkers aims to find measurable biological signs such as subtle immune activation, changes in gut permeability (6) (how easily substances pass through the gut lining), or specific chemical signatures produced by gut bacteria. Alongside this, microbiome analysis examines the patterns of bacteria and their metabolites in the gut, looking for imbalances or particular profiles linked to symptoms by comparing to current profiles of microbiomes of other individuals with diagnosed DGBIs.(7)
Another promising approach is neuroimaging, which uses brain scans to explore how communication between the gut and brain may be altered. Researchers introduce specific sensations or stimuli to the gut and then use MRI scans to observe how different regions of the brain respond. By comparing these brain responses in patients with DGBIs to those in healthy individuals, they can identify differences in brain activity that suggest altered signalling pathways. This helps explain how these changes in gut-brain communication may contribute to symptoms and affect gut function.(8)
On a more practical level, digital tools and symptom tracking apps allow patients to record real-time data on their symptoms, diet, and stress levels, helping researchers and clinicians understand symptom triggers and patterns more accurately. Ultimately, many experts believe the best path forward is through integrated models that combine psychological, physiological, microbial, and lifestyle data to create personalised risk profiles and treatment plans. These advances offer hope that, in the future, DGBIs may be identified earlier and more precisely, moving beyond diagnosis by exclusion to a more personalised and effective approach, similar to how conditions like coeliac disease are diagnosed through specific antibody and genetic tests.
Coeliac Disease is a different autoimmune disorder triggered by ingestion of gluten (a protein found in wheat, barley, and rye) in genetically predisposed individuals. Exposure to gluten initiates an immune-mediated attack on the small intestine’s lining, leading to villous atrophy and malabsorption of nutrients. Symptoms range from gastrointestinal issues like diarrhoea, bloating, and abdominal pain, to systemic effects such as anemia, osteoporosis, and neurological symptoms. Importantly, coeliac disease is strongly linked with psychiatric conditions including anxiety and depression, possibly due to chronic inflammation, nutrient deficiencies (e.g., B vitamins), and the gut-brain axis disruption caused by intestinal damage and immune activation. Strict adherence to a gluten-free diet typically restores gut integrity and alleviates symptoms but requires lifelong commitment.(9)
Disorders of Gut-Brain Interaction (DGBIs), formerly known as functional gastrointestinal disorders, are conditions characterised by chronic digestive symptoms without obvious structural abnormalities. Interestingly DGBIs are more prevalent in certain populations, including women.(10)
The most prevalent DGBI is Irritable Bowel Syndrome (IBS), affecting 10–15% of the population worldwide.(11)
IBS manifests as abdominal pain, bloating, and altered bowel habits (diarrhoea, constipation, or both), linked to dysregulated gut motility, visceral hypersensitivity, and altered gut-brain signaling. Stress and psychological factors exacerbate symptoms, reinforcing the bi-directional nature of the gut-brain axis in these disorders.(12)
Other DGBIs include functional dyspepsia (reoccuring pain in abdomen especially after eating), functional constipation, and functional diarrhoea. Treatments often combine dietary changes, pharmacotherapy, and psychological interventions such as cognitive behavioral therapy (CBT) or gut-directed hypnotherapy.
Food Intolerances and Sensitivities, such as lactose intolerance, fructose malabsorption, and non-coeliac gluten sensitivity, also disrupt gut function by impairing digestion or provoking immune responses. Lactose intolerance results from lactase deficiency, leading to fermentation of undigested lactose in the colon and causing gas, bloating, and diarrhoea. Fructose malabsorption involves incomplete absorption of fructose in the small intestine, similarly resulting in gastrointestinal symptoms. Non-coeliac gluten sensitivity is characterised by symptoms triggered by gluten ingestion without the autoimmune markers of coeliac disease. These intolerances can cause chronic gut inflammation and discomfort, further affecting mental well-being through gut-brain pathways.(13)
Scientists have many theories for how allergies manifest. One theory is that if we fail to break down a protein, it may - instead of passing through into the bloodstream - pass into the lymphatic system embedded in fat droplets causing the vigilant immune cells to attack it as a foreign body. Next time they encounter this protein, they will attack more aggressively causing allergic reactions to grow in severity e.g. the throat closing up. Another theory is that the wall of our gut can occasionally become porous, allowing food remains to enter gut tissue and bloodstream. This theory supports wheat sensitivities but both theories remain under scrutiny from researchers.(14)
Overall, these disorders show how gut dysfunction can profoundly affect both gastrointestinal health and mental health through complex interactions within the gut-brain axis. Understanding their distinct mechanisms is essential for targeted treatment strategies that address both body and mind.
1- Improving Gut and Mental Health of Those with DGBIs:
Given the central role of the gut-brain axis in DGBIs, psychological interventions have shown significant effectiveness in alleviating both digestive symptoms and mental health comorbidities such as anxiety and depression. Cognitive Behavioural Therapy (CBT) is the most extensively studied psychological therapy in this context and we see, through clinical trials, how it improves both physical and mental wellbeing.(15)
Hypnosis is a slightly newer form of therapy shown to be effective in the easing of symptoms in those with DGBIs. By using ‘thought journeys’ and ‘guided imagery’, the intensity of pain signals is reduced and these methods help alter the way the brain processes certain stimuli. For children with IBS hypnotherapy has been shown to produce a 90% reduction in pain, compared to 40% by drugs.(16)

Fecal Microbiota Transplantation (FMT) involves transferring stool from a healthy donor to a patient’s gut to restore a balanced microbiome. While FMT is well established for treating recurrent Clostridioides difficile infections, its application in DGBIs and associated psychiatric symptoms is still experimental but promising. Early studies suggest FMT may alleviate gut symptoms and improve quality of life by re-establishing a healthy microbial ecosystem and modulating the gut-brain axis.(20)
2 - Probiotics, Prebiotics and FMT
Emerging evidence supports the therapeutic potential of directly targeting the gut microbiota to improve both digestive and mental health.
Probiotics: Probiotic bacteria are helpful bacteria that positively contribute to our gut microbiome and its functions.
There are probiotic bacteria that take care of our gut by producing small fatty acids like butyrate that soothe and pamper villi. Pampered villi are more stable and more likely to grow bigger. This results in them being better at absorbing nutrients, vitamins and minerals so less harmful substances pass through. There are also probiotic bacteria that defend our gut by producing small amounts of antibiotics, acids or even by taking food away from bad bacteria. Additionally, there’s bacteria that work with the gut and immune cells to coordinate activity and response to stimuli, like how much protective mucus is needed for the intestinal lining, or what a foreign bacteria’s cell walls look like for the immune cells to provide a response accordingly.(17)
These live microorganisms, when administered in adequate amounts and in specific combinations, show hope of improving IBS symptoms. Clinical trials in IBS patients have shown that certain probiotic strains can reduce symptoms like bloating, pain, and other universal IBS symptoms.(18)
Prebiotics: prebiotics are compounds found in plant fiber which feed probiotics allowing them to grow. A new study suggests prebiotics have a role in the gut-brain axis beyond being a food source and eating them might help people choose healthier foods. Researchers recruited 59 overweight adults who regularly followed a Western diet (higher in red meat, saturated fat, and processed foods) and assigned them randomly to take either 30 grams of inulin (a prebiotic found in onions, leeks, artichokes, and bananas) or a placebo supplement, every day for two weeks. Next, the participants had brain MRIs while they viewed pictures of various low-, medium-, and high-calorie foods and indicated how much they wanted them on a one to 10 scale. The MRIs measured how much of the brain's reward network was activated when the participants made their choices. Over a second two-week period, the process was repeated: everyone took the supplement they had not taken earlier, and then underwent the MRI session. The researchers found that after taking the prebiotic supplement, people were more likely to select medium- or low-calorie foods than high-calorie ones, and their MRIs showed less activation in their brain's reward network when shown high-calorie foods.(19)
3 - Diet
Dietary interventions, particularly the low FODMAP diet (which restricts fermentable oligosaccharides, disaccharides, monosaccharides, and polyols), have proven effective in reducing IBS symptoms by minimising fermentable substrates for gas-producing bacteria, which therefore decreases bloating and pain. Diet also indirectly impacts mental health by reducing gut inflammation and restoring microbial diversity.(21)
However, while eliminating very fatty and processed foods may improve gastrointestinal symptoms when you have a DGBI, it is hard to sustain such severe changes in diet to control symptoms and when it is done too strictly it can lead to other conditions such as feeding difficulties from avoidant restrictive food intake disorder.(22)
A controlled trial called ‘A diet low in FODMAPs reduces symptoms of irritable bowel syndrome’, led by Emma P Halmos, Victoria A Power, Susan J Shepherd, Peter R Gibson and Jane G Muir shows how this low fodmap diet positively impacted IBS symptoms. They had 30 patients with IBS and 8 healthy individuals randomly were assigned to groups that received 21 days of either a diet low in FODMAPs or a typical Australian diet, followed by a washout period of at least 21 days, before crossing over to the alternate diet. Stools were then collected over days 17-21 and were assessed for frequency, weight, water content and King’s Stool Chart rating. Subjects with IBS had lower overall gastrointestinal symptom scores compared with the Australian diet and the subjects' habitual diet. Bloating, pain, and passage of wind also were reduced while IBS patients were on the low-FODMAP diet. However, symptoms were minimal and unaltered by either diet among controls.(23)





4 - Medication
Medication-based approaches typically use medications that have been used to treat conditions like depression, neuropathy, and fibromyalgia. Sometimes doctors do not clearly explain the rationale for using such medications; however, they are meant to improve the gut-brain interaction by improving pain sensation pathways in the brain, and perhaps even stimulating improved nerve function.
5 - Suplements
Sometimes people consider acupuncture, ginger, turmeric, or herbal supplements, which you should always discuss with your doctor to prevent side effects or serious health issues, including liver problems.(24)


6 - Fermented Foods
Many foods that are fermented undergo lacto-fermentation, in which natural bacteria feed on the sugar and starch in the food, creating lactic acid. Not only does this process remove simple sugars, it creates various species of good bacteria, such as Lactobacillus or Bifidobacterium. The exact amounts and specific strains of bacteria in fermented foods vary depending on how they are made. In addition to probiotics, fermented foods may contain other valuable nutrients like enzymes, B vitamins, and omega-3 fatty acids.(25)
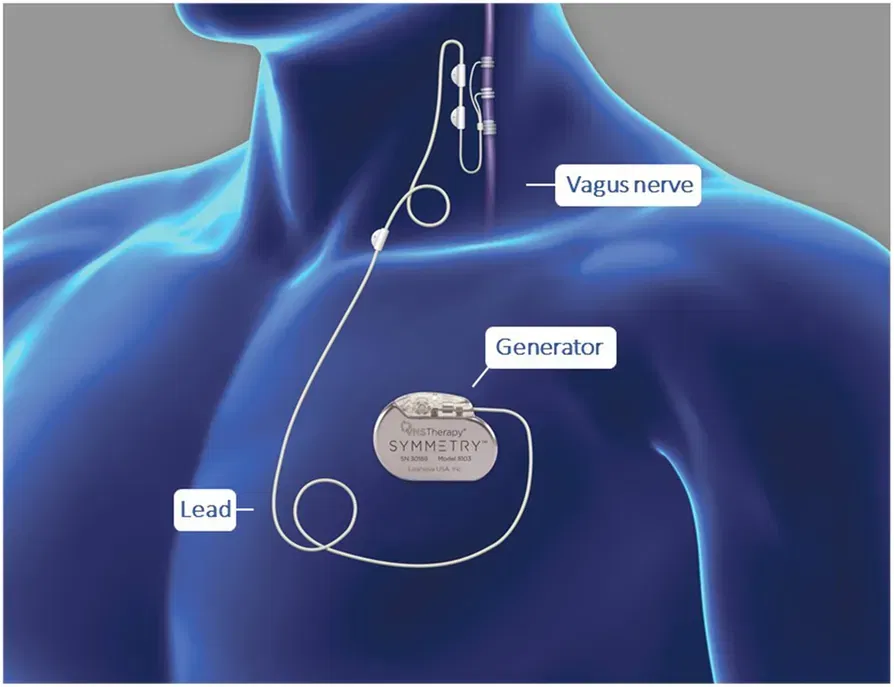
7 - Vargus Nerve Therapy
The vagovagal reflex is a neural circuit in which sensory signals (afferents) travel from the stomach to the brainstem, which then sends motor signals (efferents) back to the stomach via the vagus nerve. This reflex helps regulate gastric accommodation, motility, and secretion - essential functions often disrupted in Disorders of Gut-Brain Interaction (DGBIs). Modulating vagal efference has the potential to improve gut motility, reduce visceral hypersensitivity, and even influence emotional regulation, making the vagus nerve a promising target for therapeutic intervention.
Recent research has explored non-invasive ways to influence vagal activity, such as transcutaneous auricular vagus nerve stimulation (taVNS). A recent study introduced a specialised technique called Respiratory-gated Auricular Vagal Afferent Nerve Stimulation (RAVANS), in which electrical stimulation of the ear (innervated by the auricular branch of the vagus nerve) is timed with exhalation to align with natural peaks in vagal tone. The study found that RAVANS significantly enhanced gastric accommodation in healthy individuals. While promising, this approach is still in early research stages, and its clinical effectiveness in treating DGBIs requires confirmation in larger, symptom-focused trials.(26)
Emerging Research & Controversies:
A Vicious Cycle?
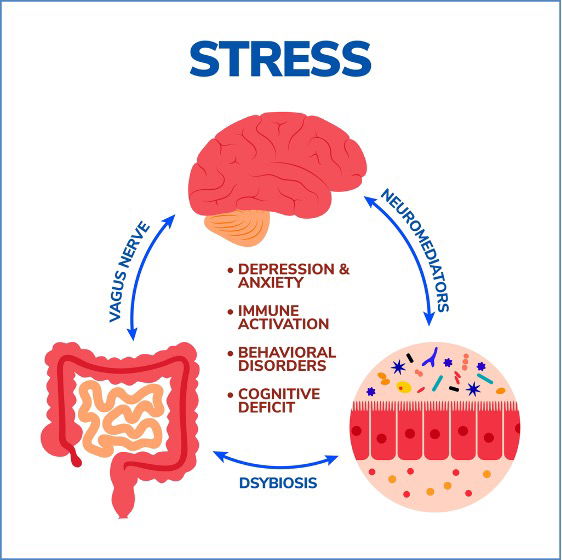
Recent advances in gut-brain axis research have illuminated complex interactions that suggest a vicious cycle may perpetuate both gastrointestinal dysfunction and mental health disorders. The interplay between gut microbiota imbalance (dysbiosis), chronic inflammation, and altered neural signaling forms a feedback loop where each factor exacerbates the others, complicating treatment and understanding.
The gut-brain axis plays a crucial role in how the body responds to stress, and recent research highlights the gut microbiota as a key regulator of this system. Diet, early-life factors, and environmental influences shape the microbiota, which communicates with the brain through pathways like the vagus nerve, the immune system, and microbial metabolites. Disruptions in the microbiota may contribute to stress-related conditions like anxiety, depression, and IBS, though much of the evidence comes from animal or correlational studies. The emerging field of psychobiotics aims to improve mental health by targeting the microbiome, but more human research is needed to confirm these effects.(27)
While some researchers argue that gut dysbiosis initiates mental health symptoms, others propose that psychological stress and brain dysfunction may primarily drive changes in the gut microbiome. The reality likely involves a bidirectional cycle where the gut and brain affect each other in significant enough amounts, that if any problem were to occur, dysbiosis and in some cases mental health problems then develop. The gut-brain axis is highly complex, and it remains uncertain whether the brain or the gut has a greater influence over the other. This dynamic makes treatment and the identification of specific triggers challenging to unravel.
Additional controversies include the clinical efficacy and safety of microbiome-based therapies, such as probiotics and fecal microbiota transplantation (FMT). While promising, these interventions yield mixed results across studies, with variable patient responses and unclear long-term effects. The diversity of microbial communities and the fact that usually various factors all contribute to the DGBI, complicates interpretation. There is also concern over potential risks, including pathogen transmission with FMT.(28)
Although there is research suggesting positive impacts in probiotic supplementation,(29) there are also concerns with this form of probiotic intake.(30)
Moreover, individual variability in genetics, diet, environment, and lifestyle further influences gut-brain axis dynamics, highlighting the need for personalized approaches. As such, current research advocates for integrative models combining microbiome science, neurobiology, immunology, and psychology to fully unravel this complex system.
Conclusion
The intricate relationship between the gut and brain challenges traditional boundaries between physical and mental health, revealing a complex, bidirectional communication system where disruptions in one can profoundly affect the other. As explored, the enteric nervous system, vagus nerve, and gut microbiota form a network that regulates digestive function, immune responses, and emotional well-being. Conditions such as coeliac disease, DGBIs and food intolerances, illustrate how gut dysfunction often coexists with psychological symptoms like anxiety and depression, showing the need for integrative treatment approaches.
Research does show that it is difficult to identify the source of triggers for DGBIs due to multiple factors contributing to the miscommunication within the gut-brain axis. Many theories are still developing and results from developing research are shaping the way we tackle gastrointestinal disorders, usually calling for a holistic and integrative model that addresses the interconnected nature of the gut and brain in order to maximise improvement in all areas of those with gut-brain interaction disorders.
References
1https://medicine.missouri.edu/news/irritable-bowel-syndrome-patients-suffer-high-rates-anxiety-and-depression
2https://www.frontiersin.org/journals/immunology/articles/10.3389/fimmu.2019.00277/full
3https://www.josam.org/josam/article/view/90#title-4
4
5 https://pubmed.ncbi.nlm.nih.gov/17183312/
6 https://pubmed.ncbi.nlm.nih.gov/34009040/
7 https://pmc.ncbi.nlm.nih.gov/articles/PMC6391518/#S6
8 https://pmc.ncbi.nlm.nih.gov/articles/PMC6999847/#S27
9 https://pubmed.ncbi.nlm.nih.gov/28760445/
10https://www.health.harvard.edu/blog/recognizing-and-treating-disorders-of-gut-brain-interaction-202204202730
11 https://www.cghjournal.org/article/S1542-3565(12)00308-4/
12https://www.gastrojournal.org/article/S0016-5085(16)00223-7/
13 https://www.mdpi.com/2072-6643/9/11/1268
14 ‘Gut’ book by Giulia Enders pg 53-54
15https://pubmed.ncbi.nlm.nih.gov/27870997/
16https://www.gastrojournal.org/article/S0016-5085(07)01629-0/
17 ‘Gut’ book by Giulia Enders page 230
18 https://onlinelibrary.wiley.com/doi/10.1111/apt.15001
19 https://pubmed.ncbi.nlm.nih.gov/37793780/
20 https://pubmed.ncbi.nlm.nih.gov/29980607/
21 https://pmc.ncbi.nlm.nih.gov/articles/PMC8228761/?
22https://www.health.harvard.edu/mind-and-mood/ask-dr-rob-about-eating-disorders#:~:text=Avoidant%2Frestrictive%20food%20intake%20disorder%20%E2%80%94%20With%20this%20condition%2C%20people,psychological%20explanation%20for%20these%20symptoms.https://www.health.harvard.edu/mind-and-mood/ask-dr-rob-about-eating-disorders#:~:text=Avoidant%2Frestrictive%20food%20intake%20disorder%20%E2%80%94%20With%20this%20condition%2C%20people,psychological%20explanation%20for%20these%20symptoms.
23 https://pubmed.ncbi.nlm.nih.gov/24076059/
24 https://pmc.ncbi.nlm.nih.gov/articles/PMC5502701/
25 https://www.ncbi.nlm.nih.gov/books/NBK234703/#ddd00072
26 https://faseb.onlinelibrary.wiley.com/doi/abs/10.1096/fasebj.2020.34.s1.02939
27 https://pubmed.ncbi.nlm.nih.gov/29276734/
28 https://pubmed.ncbi.nlm.nih.gov/31578306/
29 https://onlinelibrary.wiley.com/doi/full/10.1111/nmo.14751?
30 https://pubmed.ncbi.nlm.nih.gov/31061539/
Amanda Dias © Gut Instincts 2025. All rights reserved.